The quality of your edible cannabis product can only be as reliable as the components that comprise it. The three types of components include active ingredients (such as CBD oil), packaging components (such as the bottles that hold finished product) and inactive ingredients (such as coconut oil). When evaluating a potential supplier for these three areas, a risk-based method follows a vendor selection process that highlights critical ingredients and also adequately assesses excipients. With this approach, the vetting process for a supplier is based on the impact the potential ingredient or component will have on the quality and purity of the finished product.
Choose only those suppliers who can provide certification that the packaging components are food-grade or food-safeThere are three basic categories to guide vendor assessment. Is the supplier providing 1) a packaging component with product contact, 2) an excipient, or inactive ingredient, or 3) the active ingredient? Regardless of the category, due to the factious nature of cannabis, it is important to first verify with a vendor that it will sell its products to a company in the cannabis industry. Once that is determined, the evaluation process may begin.
Packaging Components

Packaging components, such as bottles and caps, are considered primary packaging because they have direct contact with the finished product. Suppliers of the primary packaging must be able to provide assurance that their goods do not contain additives that are harmful to consumers. Therefore, choose only those suppliers who can provide certification that the packaging components are food-grade or food-safe. Reputable vendors will also be able to provide a certificate of compliance, also known as a certificate of conformance, which states that the component meets specifications required for that part. Many cannabis regulations require finished products to be sold in child-resistant packaging, so the supplier will need to provide child-resistant certification for the packaging components, if applicable.
Excipients
Excipients are ingredients that are added to a product for the purposes of streamlining the manufacturing process and enhancing physical characteristics such as taste and color. Some examples could include coconut oil, starch and alcohol. Though they do not have the same critical nature as active ingredients, their potential risk to a finished product is generally greater than that of a packaging component. As such, there are additional factors to consider for an excipient vendor. Verify with the supplier that it can provide the following documentation. While governing regulations may not require some information, the data included in these documents are important to ensure the quality of your finished product.
- Certificate of Analysis (or, certificate of conformance), for each lot of material. The information on a certificate, including the tests performed, specifications and test results must be sufficient to determine if the material is acceptable for use in the product.
- Allergen Statement. This statement is important to accurately include or disclaim allergens on the finished product label.
- Residual Solvent Statement. Solvents are commonly used to bolster the manufacturing process for a material. In order to maintain acceptable levels of residual solvents in a final product, it is necessary to also consider the toxicity and level of each solvent in the raw material.
- Heavy Metals Certification. Since metals pose a risk to consumer safety, it is important to know what amounts, if any, are being contributed to your product by raw materials.
Because changes in an excipient can impact your finished product, make every attempt to obtain a commitment from a supplier to notify you if changes are made to the excipient’s specifications.
Active Ingredients
Cannabis oil is the ingredient that, when the edible cannabis product is consumed, is biologically “active.” Thus, it is considered to be the active ingredient in cannabis products. Since cannabis oil has a direct impact on the quality of a product, it is critical that the oil supplier be appropriately evaluated. One of the main considerations for a cannabis oil supplier is whether the supplier is willing to host initial and periodic audits of its manufacturing facility. Such audits are crucial in assessing the capability of the vendor to comply with regulatory requirements and established procedures – can the supplier consistently provide quality material? The answer to this question is too important to risk for you and your customers.As anyone working in the industry has experienced, anything related to cannabis is placed under an unprecedented critical lens.
Additionally, verify the oil supplier will provide key documentation, such as that listed above for excipients, to support the quality and purity of the oil. And last but not least, ensure the information reported by the supplier is adequate to meet the requirements of your finished product.
Evaluation guidelines and criteria such as these should be added to standard operating procedures to ensure consistency and quality across all products. As anyone working in the industry has experienced, anything related to cannabis is placed under an unprecedented critical lens. The importance of consumer safety and bolstering industry integrity is paramount. Sourcing validation is a critical initial step in the production process that can directly impact a company’s success and longevity in the cannabis industry.








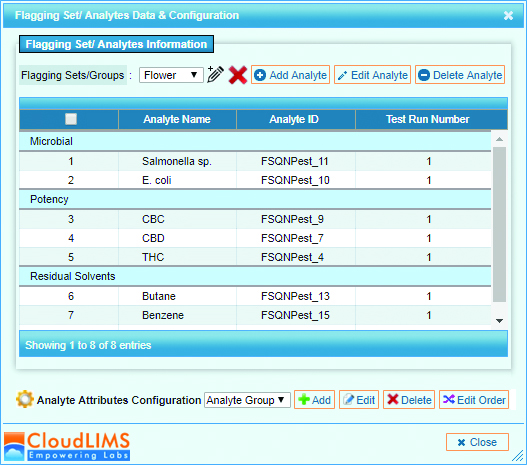
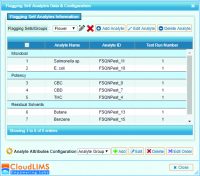

















 Each lab has access to raw, anonymized data including a consensus mean, z-scores and kernel density plots. This round measured how well 35 cannabis labs perform in testing for potency, pesticides, residual solvents and microbial contaminants such as E. coli, Salmonella, Coliform, yeast and mold.
Each lab has access to raw, anonymized data including a consensus mean, z-scores and kernel density plots. This round measured how well 35 cannabis labs perform in testing for potency, pesticides, residual solvents and microbial contaminants such as E. coli, Salmonella, Coliform, yeast and mold.




